Cruciferous or Brassica vegetables, like broccoli, have long been promoted as a key part of a healthy diet—and rightfully so. The veggies in this family have been associated with reduced risk of heart disease and stroke, [1] and are being studied for their potential role in cancer prevention. [2] But beyond broccoli, at least 40 types of cruciferous vegetables are consumed worldwide, including bok choy, cabbage, cauliflower, collard greens, and kale, as well as zesty turnips, radishes, arugula, and mustard greens.
If the mention of these vegetables spark any distinct, olfactory-related memories of overcooked greens, you’ll be interested to know that cruciferous vegetables owe their unique smell and somewhat bitter flavor (to which some may be more sensitive than others) to the breakdown of sulfur-containing phytochemicals called glucosinolates. However, brassicas don’t have to be destined to an over-boiled and bitter fate! While  some are delicious left raw, simple preparation techniques such as roasting or sautéing can bring a natural sweetness to the forefront.
some are delicious left raw, simple preparation techniques such as roasting or sautéing can bring a natural sweetness to the forefront.
To see some culinary chemistry in action, we visited the kitchen of Guy Crosby, Adjunct Associate Professor in the Department of Nutrition, as he discussed the science of cooking and prepared a staple at many holiday tables—Brussels sprouts.

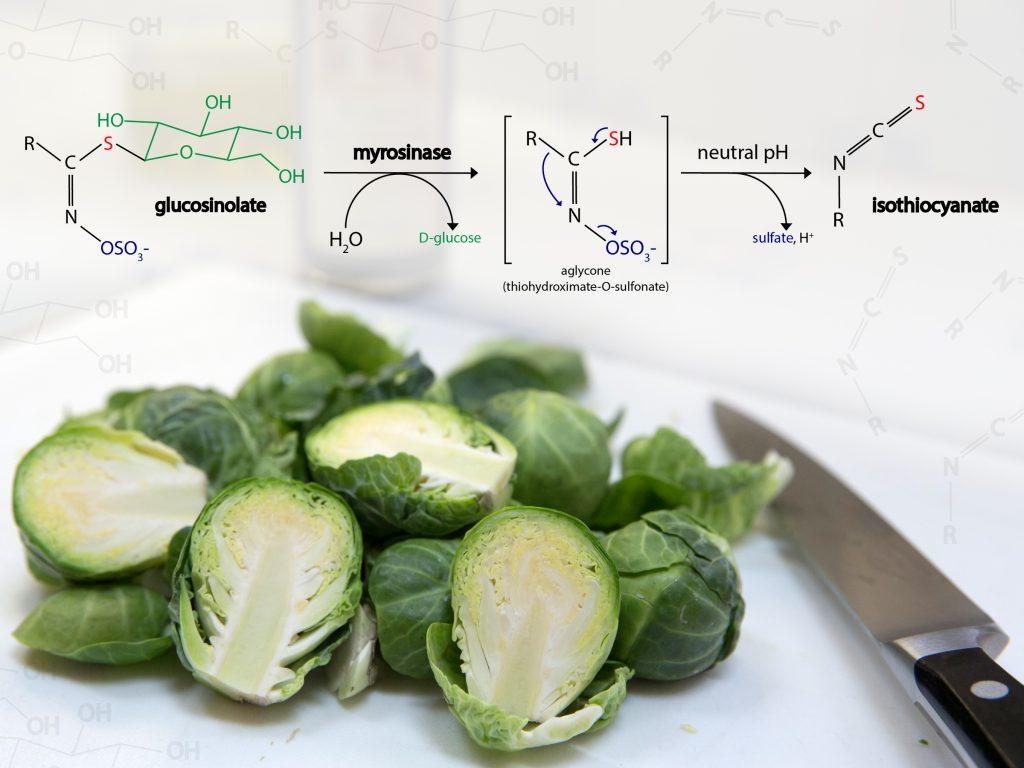
Cruciferous vegetables like Brussels sprouts are full of glucosinolates, sulfur-containing compounds responsible for creating their strong aroma and somewhat bitter flavor. But if you hold an intact Brussels sprout to your nose, you might notice it doesn’t smell much at all!

This is because the scents and flavors aren’t created until you chop, cook, or chew. Damaging the cell walls releases an enzyme (myrosinase) that breaks down glucosinolates into biologically active compounds called isothiocyanates.
Despite their bitter-tasting compounds, carefully oven-roasting Brussels sprouts can bring out a nutty, almost popcorn-flavored sweetness.

With just a bit of healthy fat like olive oil and some salt, the dry heat will brown the sprouts, thanks to a process called the Maillard Reaction. When food is heated above 300F, browning occurs due to this chemical reaction between amino acids and certain sugars (called “reducing sugars”), like glucose and fructose. Beyond the change in color, the Maillard Reaction is also responsible for those enticing smells of baking and roasting—as well as the resulting transformed flavor of the food.

Simple Roasting Instructions: Halve the Brussels sprouts and arrange cut-side-down in a single layer on a baking sheet lined with parchment paper or in a baking dish. Drizzle with olive oil and pinch of salt. Roast in a preheated oven at 400 F until browned on the exterior and tender on the inside. Smaller Brussels sprouts (about 1” in diameter) should be roasted for 18 minutes, while those 1.5” in diameter or larger may take 20-25 minutes. Serve with a splash of balsamic vinegar or fresh squeezed lemon juice.
Related
- Learn more about Brussels sprouts, as well as other preparation methods and serving suggestions, including roasted Brussels sprout “chips.”
- Listen to Guy Crosby explain how you can use food science to cook the perfect holiday meal – Harvard Chan: This Week in Health, Science of Thanksgiving
[soundcloud url=”https://api.soundcloud.com/tracks/294108599″ params=”auto_play=false&hide_related=false&show_comments=true&show_user=true&show_reposts=false&visual=true” width=”100%” height=”450″ iframe=”true” /]
References
- Hung, H.C., et al., Fruit and vegetable intake and risk of major chronic disease. J Natl Cancer Inst, 2004. 96(21): p. 1577-84.
- Wiseman, M., The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: a global perspective. Proc Nutr Soc, 2008. 67(3): p. 253-6.

